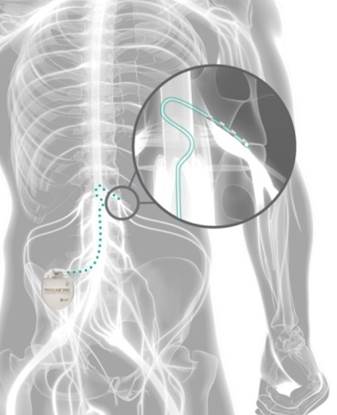
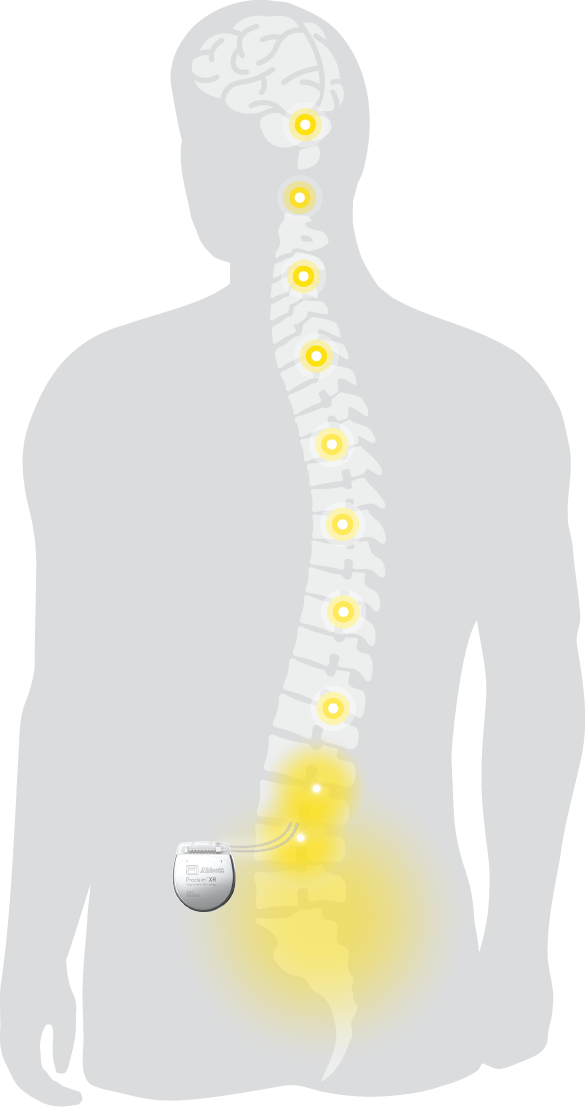
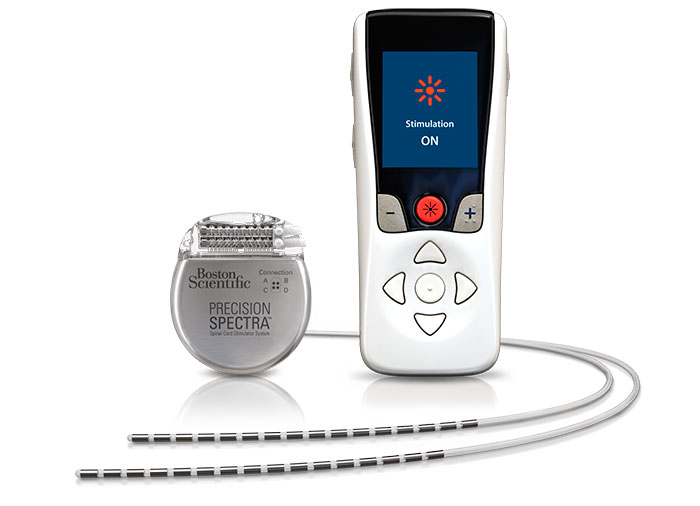
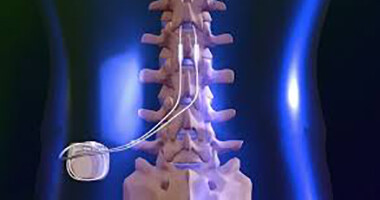
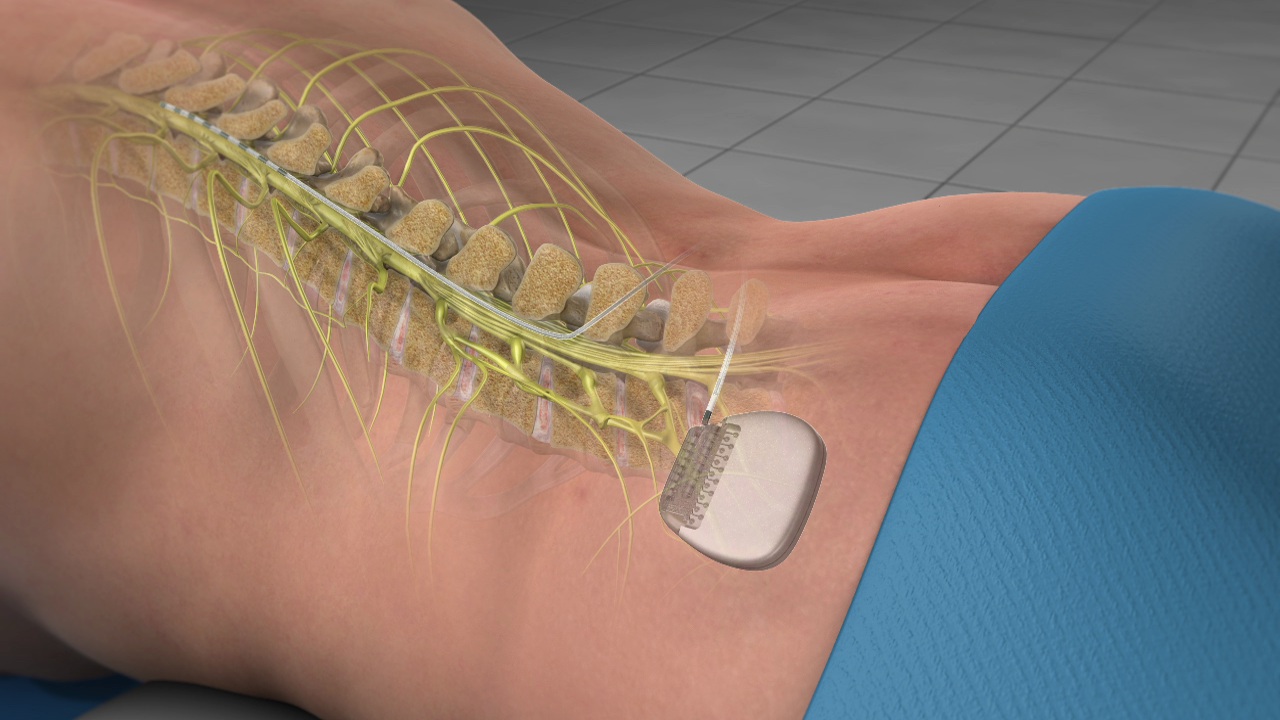
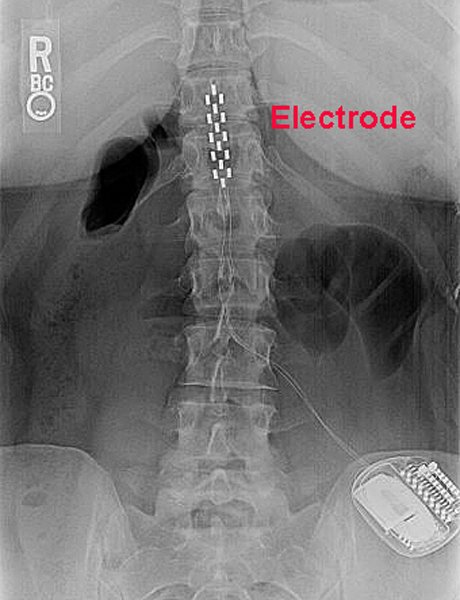
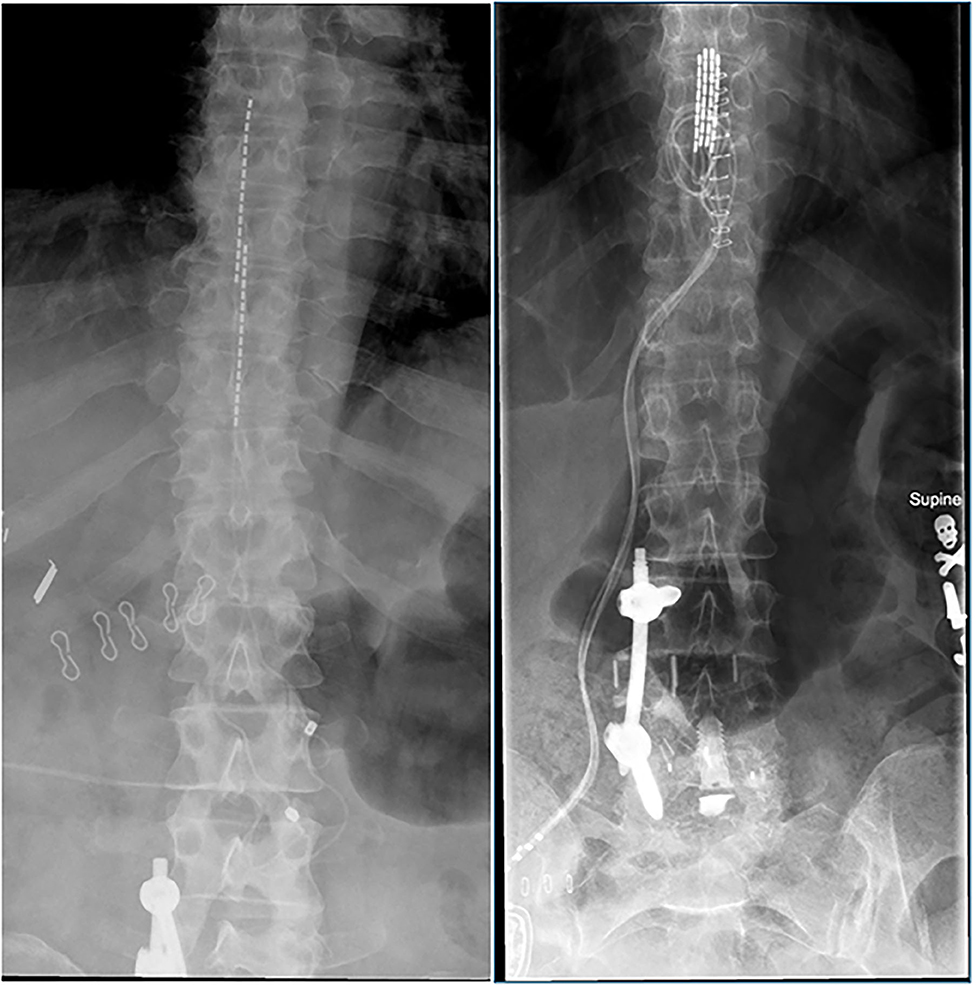
FDA Approves Abbott's "Low Dose," Recharge-Free Spinal Cord Stimulation System with up to Ten Year Battery Life* for People Living with Chronic Pain - Sep 26, 2019
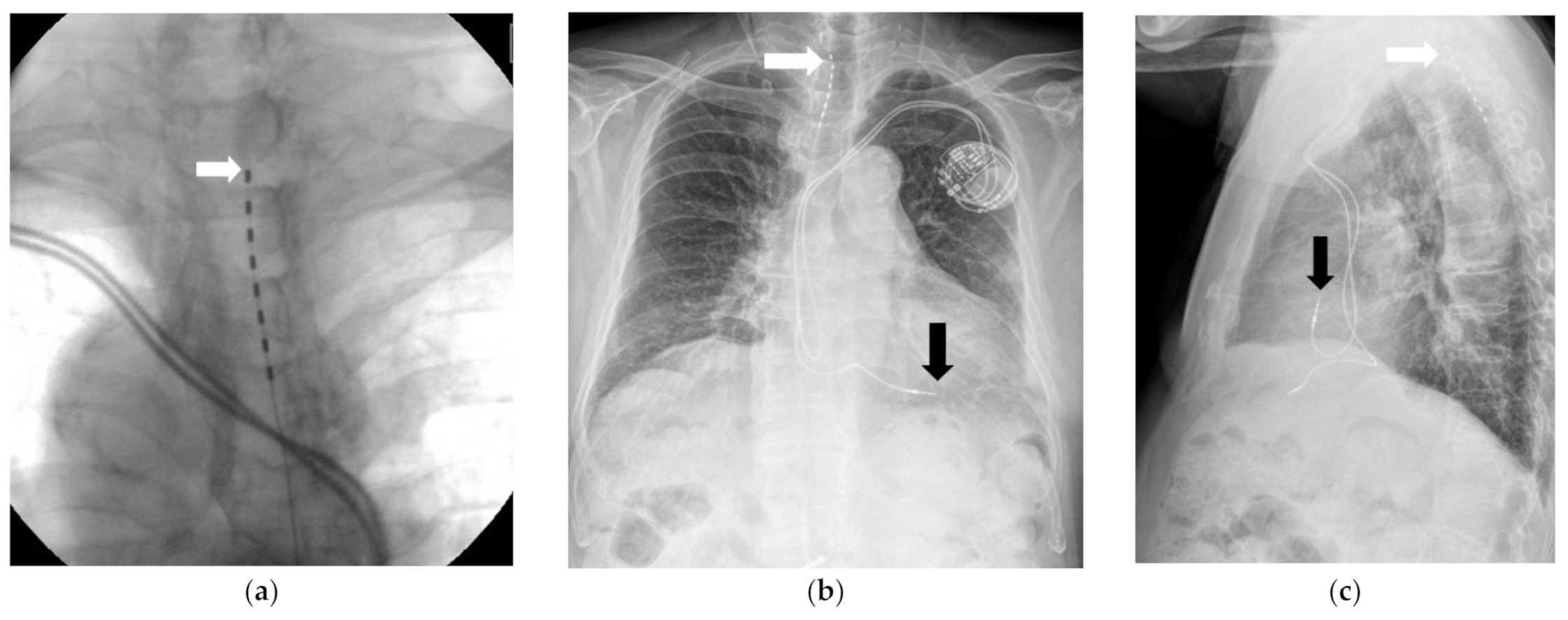
Medicina | Free Full-Text | Burst Stimulation of the Thoracic Spinal Cord near a Cardiac Pacemaker in an Elderly Patient with Postherpetic Neuralgia: A Case Report
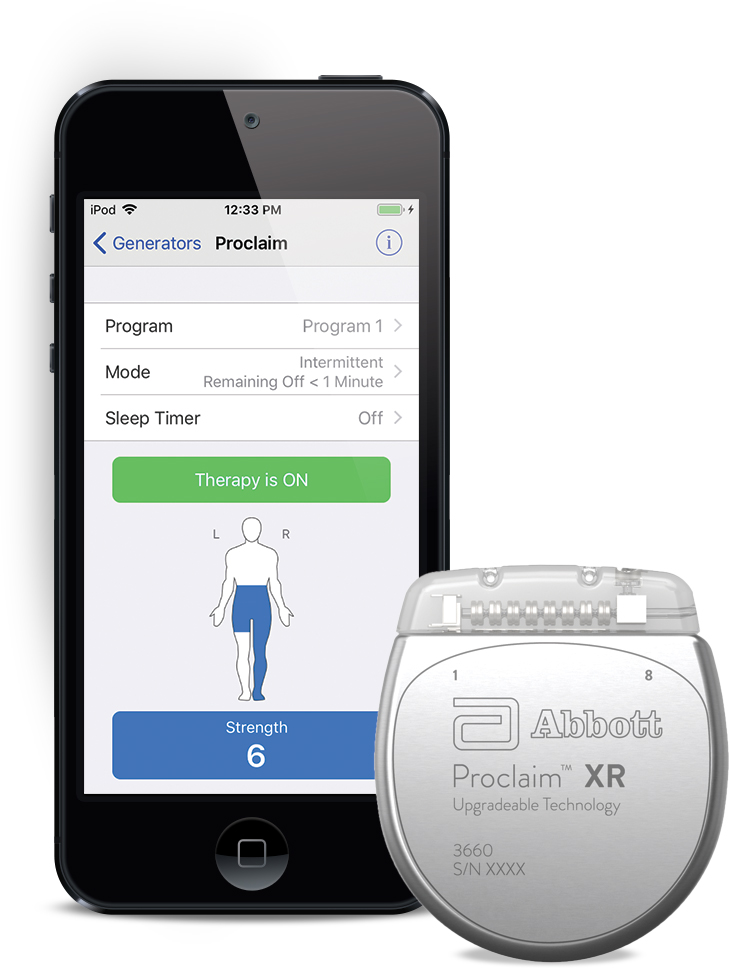
FDA Approves Abbott's "Low Dose," Recharge-Free Spinal Cord Stimulation System with up to Ten Year Battery Life* for People Living with Chronic Pain - Sep 26, 2019